 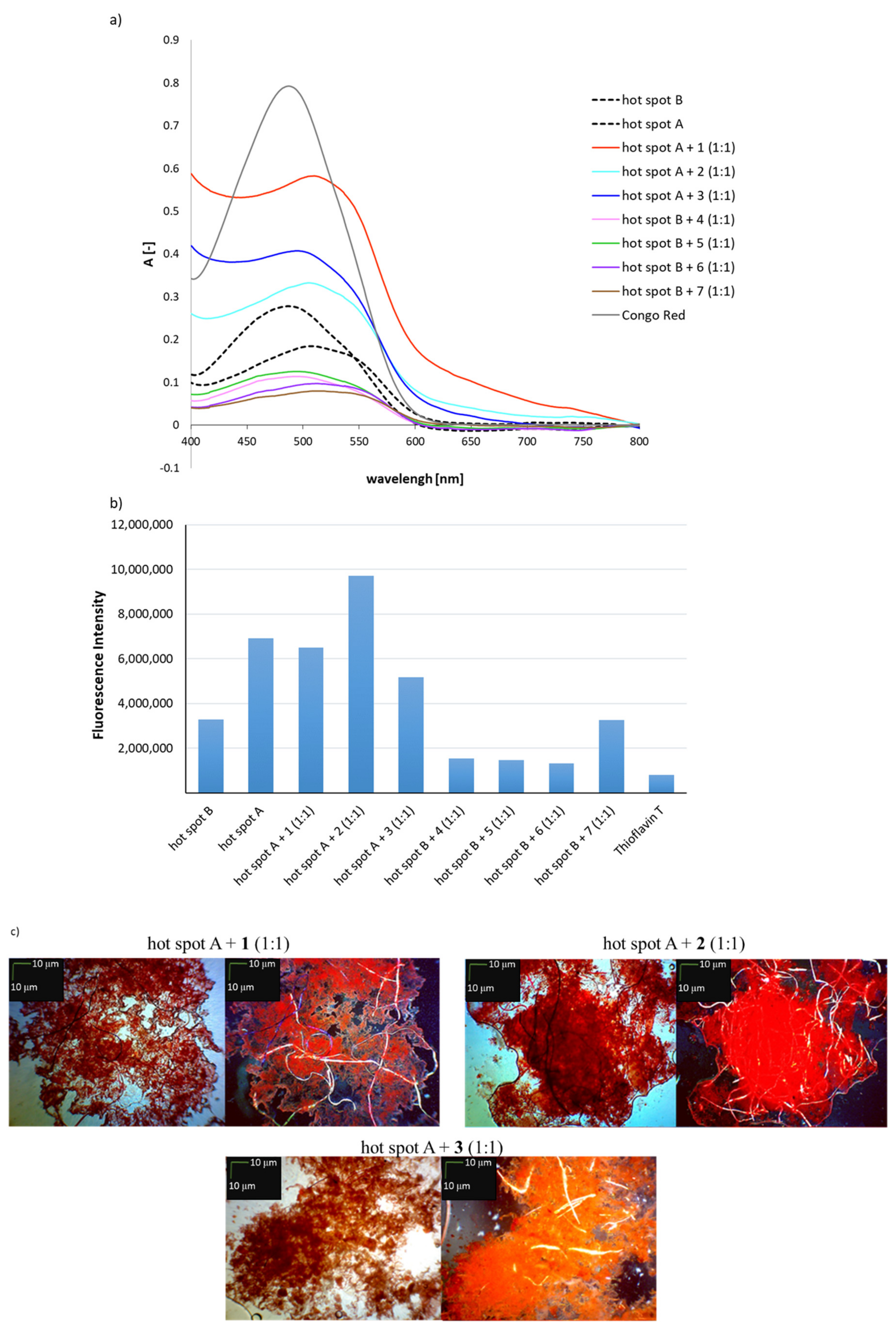
The hallmark of AL amyloidosis is the transformation of soluble monomeric protein into insoluble amyloid fibrils 12, 13. N-terminal fragments, comprising mostly the variable domain (V L) of the LC often represent the amyloidogenic species. Furthermore, the circulating free LCs often undergo proteolytic cleavage prior to assembly into insoluble amyloid fibrils 8, 9, 10, 11. Patient-specific mutations in the LC, which destabilize the native fold and consequently trigger fibril formation are an important element of the disease 4, 5, 6, 7. The disease is caused by an underlying plasma cell dyscrasia that entails the elevated expression and secretion of free antibody light chains (LC) 2, 3. Light chain (AL) amyloidosis is the most common type of systemic amyloidosis 1. Together, our results reveal a rate-limiting multi-step mechanism of structural transitions prior to fibril formation in AL amyloidosis, which offers, in the long run, opportunities for therapeutic intervention. Structural transitions from an anti-parallel to a parallel β-sheet secondary structure occur in the oligomers prior to amyloid formation. During oligomerization, the hydrophobic core of the LC domain rearranges which leads to changes in solvent accessibility and rigidity. This is a prerequisite for the formation of an ensemble of oligomers, which are the precursors of fibrils. 
The process starts with partial unfolding of the V L domain and the formation of small amounts of dimers. We have dissected the lag phase of a patient-derived LC truncation and identified structural transitions that precede fibril formation. The events occurring during this lag phase are largely unknown. In vitro, AL fibril formation is a slow process, characterized by a pronounced lag phase. In antibody light chain (AL) amyloidosis, overproduced light chain (LC) fragments accumulate as fibrils in organs and tissues of patients. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
January 2023
Categories |
 RSS Feed
RSS Feed
